Rebecca Giggs is an award-winning author from Perth, Australia, who writes about how people feel toward animals in a time of ecological crisis and technological change. Her work has appeared in Granta, The Atlantic, The New York Times Magazine, Best Australian Science Writing, and other publications. Her first book, Fathoms: The World in the Whale, was awarded the Andrew Carnegie Medal for Excellence in Nonfiction and was a finalist for the Kirkus Prize and the PEN/E.O. Wilson Literary Science Writing Award.
Studio Airport, founded by Bram Broerse and Maurits Wouters, is an interdisciplinary design studio that ventures out into the cultural ether to forage for anomalies, creating work that spans art, culture, science, and ecology. In addition to Emergence Magazine, their creative partners include the Design Museum, See All This Art Magazine, Slowness, Normal Phenomena of Life, and Sapiens Magazine. They serve as master tutors at the Design Academy Eindhoven and were recognized as European Agency of the Year 2024 by the EDA.
Very small beings are often responsible for vast surges of life. Rebecca Giggs follows the mass migration of the bogong moth in alpine Australia: a story of superabundance and apocalypse.
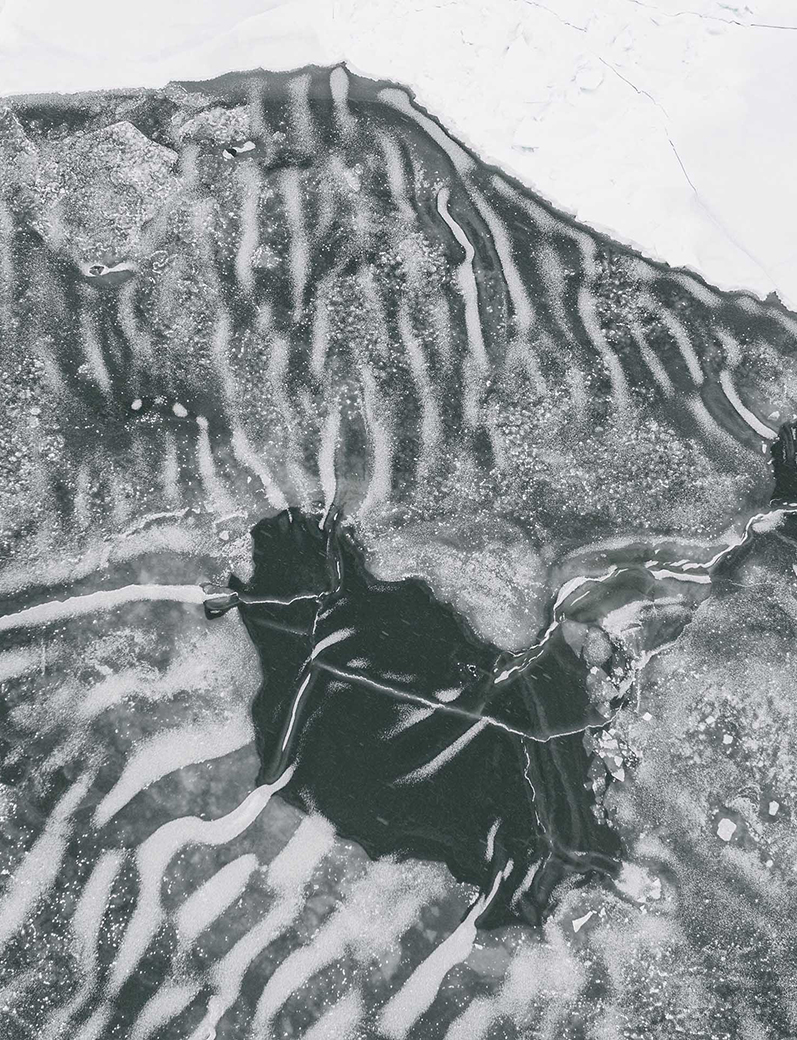
The moths, when they came, were said to appear first like sea fog massing above the ocean. Lighthouse keepers along the southeastern edge of Australia warned of beacons so darkly swarmed that on-water navigators doubted their bearings. Ferryboats were burdened by thousands, wings ablur. Some moths hung in clusters off the precipitous coastal cliffs, living icicles, dripping with more moths. The moths, as a myriad, moved in: at nightfall they “swept over the suburbs in clouds,” wrote one reporter for a Sydney tabloid, The Sun. Descending into tea trees and turpentine gums in Gosford, the seething of the moths gave the impression of bough-shaking winds when all else was motionless.
If the moths’ light-seeking caused disruption in the darkness, their urge to seek shelter when the dawn broke made them a more invidious presence yet. So many of their oily bodies were crushed on train tracks that slowdowns were mandated to stop locomotives slipping from the rails. They jammed the circuitry of elevators, spoiled gatherings. At a garden party at Government House in the inland capital of Canberra, every iced cake was seen to be decorated with moths. The moths entered people’s houses. They crept behind upright pianos, into radio sets, betwixt the slats of venetian blinds. They got between the mattress and bedsheets, and huddled in the pockets of dress suits. In kitchens, gutted fish were found to have bellyfuls of moths. If a light was switched off, hundreds of tiny arrow shapes might fan out from beneath paintings hung on the wall. One year, churchgoers counted eighty thousand moths on the windows of Saint Thomas’s prayerhouse in North Sydney. Services were canceled for seven days, the building sealed while the moths congregated under the eaves. People reached for words like visitation, marvel. The less-enraptured said: plague.
These were migratory moths, called bogong moths, and through the early twentieth century few people could say with confidence where they came from. “Noiseless messengers,” The Argus newspaper deemed the bogongs in 1916—“noiseless messengers sent forth to flicker ghost-like through space, and collect the news of other worlds.” Truth was, the moths had their origins underground. After frail frosts, and when the early spring was wet, great throngs of moths emerged from pupae in the soils of lowland southern Queensland and in western and northwestern New South Wales. Stirred by some ephemeral cue (temperature, day length, barometric pressure), the moths took off. Though no single individual in their generation had ever made the journey—and while each moth’s brain is scarcely a speck—the moths, their bearings imbued with instinct, set out to travel over one thousand kilometers (620 miles).
Some years were sparser than others, but when conditions favored the moths, there could be over four billion on the wing. Passing through the railyards of Newcastle, they obscured electric signals. As far south as Mirboo North in Gippsland, men complained of needing to move agglomerations of moths off the paddocks by the shovelful. Lacking the necessary mouthparts to chew leaves, they did not skeletonize plant life as locusts do. Instead, the moths relied on floral sugars to power them, supping thin streams of nectar via their proboscises, along with lerp—a type of honeydew extruded by louses. Each moth could only ingest a skerrick of sweetness, but they were so numerous that apiarists nonetheless found they had to sustain their bees on syrup after the moth front had passed by, taking with it much of the nourishment otherwise found in flowering yellow box, red box, grevillea.
Many moths were killed—by nightjars and frogmouths, by high winds, by sizzling up in light fixtures, and by slapping hands—but there always seemed to be more to come. They were impervious to knock-down sprays. Any attempt at sweeping them from a surface left behind black pencil marks. In Dubbo 1919: the moths “destroy[ed] the happiness of many a domestic circle, and by their dying help[ed] to increase the cost of living.” Removing moths from the home was nearly impossible. One might as soon have tried to net a mist and tow it back out to sea.
Yet, not that long after they arrived, the moths disappeared from the cities, like a nightmare dissipated on waking. Where had the moths gone to? From at least the end of the last ice age, the moths have taken their leave, every year, to go into hiding in the Australian Alps. The bogongs chase the cold. Their metapopulation, which has several reservoirs inland of the Dividing Range, funnels together to seek refuge from the hottest weather of summer by climbing up above the tree line into chilled crevices and grottoes in the high-altitude scree of the Snowy Mountains, the Victorian Alps, and the Brindabellas. When they finally enter their encampments in the granite and basalt, the moths settle on rock faces in a tessellating pattern. Each moth, a jigsaw piece, tucks its head under the hindwings of the one before it, until there is a wide brocade of moths that can extend for maybe eighty meters squared, or more. If they blanket the interior of a cave, it can come to feel like a softly padded cell. The moths enter a torpor called “aestivation.” Aestivation is the opposite of hibernation: it is done to circumvent the swelter, not the snow. The moths are mostly motionless. Intermittently they jiggle their wings. A handful might take a turn in the open air each night, moving as on an orrery, before settling into their long tranquility again. Bogongs live much longer than the average moth: between eight and nine months. They will stay there, in the icy dark, across the turn of the new year, before returning to breed, lay eggs, and die where they were born, in the cracking clays far away.
The Jaithmathang, Gunaikurnai, and Taungurung peoples knew of this migration—and had known since long before European invasion. The moths’ name, “bogong,” comes from the southeastern Indigenous language groups. Bungung denotes a moth of the mountains, or the mountains of the moths, and the brown color that envelopes both. The aestivation of the moths was the incitement for a pilgrimage of Indigenous Australians into the lowlands and foothills of the Alps, land that was cyclically inhabited by the Traditional Owners who have been its continuous custodians, and in its care, for all of the timespan the Dreaming encloses. At several waypoints along the moths’ passage, people stupefied the insects with smoke and cooked them in coalless fires before grinding their bodies into a paste and fashioning long-lasting patties. For the moth hunters, bogongs were a seasonal cornerstone of their diet. The custom was so widespread that it changed the appearance of the landscape: the ground was rumpled like a quilt from where the firepits were dug year after year. Significant law, intergroup consultation, and ceremony are associated with the occupation of the high plains at moth-harvest time. Dispossession and colonial violence disturbed these practices.
For centuries the bogong moths streamed back into these caves, slept, and vanished again when the weather cooled. The hands of the moth hunters illustrated facets of the feast on rock walls. Later came scientists, from research institutions and universities, to study the moths across the tors of several summits. The scientists noted that the moths smelled sweet like molasses when they arrived, and thereafter awful, like compost. Then, in the summer of 2017/18, the bogong moths, prolific as they had been for all the years prior, vanished more completely. Where once there had been hundreds of thousands of the insects—a juggernaut, a moving nimbus—now the night air stood empty. On Mount Morgan and Mount Gingera: no moths. A cave long favored by moths on a boulder outcrop near South Ramshead, in the Kosciuszko Main Range, saw only a smattering very deep in the far reaches; likewise in known habitat on Mount Buffalo. The next summer a single live moth was found on Mount Morgan. Three moths made it to Mount Gingera. With a note of terror the scientists reported that within a few short years bogong numbers had declined by 99.5 percent. This past December the International Union for the Conservation of Nature (IUCN) formally listed the bogong moth as endangered. What has happened to the moths, and what will happen in the mountains if they are not restored, has much to tell about how we envision the manifold crises we are connected to, and the scale on which they occur.
I had begun reading about the moths at a time when we were compelled to stay indoors, to wait out a state of semi-dormancy in lockdown. It started with a dream, the dream everyone seemed to be having that spring, of a billowing swarm of insects. In mine, the insects were seen from far off—a murmuration knotting and unknotting on the horizon, banking into mountainous peaks that shivered and collapsed. At a distance I registered only curiosity towards them, but then the insects started to collect together, to concentrate, and, with clear intent, cascade down upon the buildings of the city into the streets. A horrible fear gripped me. Long minutes after waking I could still feel the prickling of legs and wings landing all over my skin.
Researchers who study dreams in the context of tragedy have observed that, since the outset of the pandemic, the imagery of insects has proliferated in sleeping minds. A virus is not a living organism, but we sometimes call it a “bug,” so the theory holds that bugs (of the arthropod kind) were a ready-made metaphor to visualize an invisible threat. It was easy to imagine an insect horde passing through people’s heads on their pillows at night, like a storm traversing the eastern seaboard. Friends detailed variations on the theme: one in which ants overtook a classroom, one where a train carriage filled with hornets. My aunt dreamt that she was compelled to hold a single insect in her mouth as she moved through a crowd. Bitter, she said.
Insects are bonded to ideas of mortality the world over, being both decomposers and natural transformers: Scarab beetles, a feature of funerary art in Ancient Egypt. Jade cicadas, placed on the tongues of the deceased in the Han Dynasty to ease a person’s transition into the afterlife. In the portraiture of pre-seventeenth-century Europe, the addition of a fly signified the subject was no longer alive. And the death’s-head hawkmoth, of course, was a harbinger of epidemic illness and pestilence in France. To be touched by insects—to be traipsed by a lacewing, or to cup a centipede in your palm—has a morbid voltage, I think, as a foretoken of the moment the body ceases to sense the lightest contact, before it begins to turn, in time, to stillness, to ash on the wind, or dust on the touchpaper texture of a moth.
After the curfew each evening, I sat at home with the lamps on, watching whatever pinwheeled and buzzed against the glass. The borders were closed and a five-kilometer travel limit had been imposed, in addition to social distancing measures. Life seemed to have contracted down to very little. The window had become my public square, I thought. Could I get interested in what was there, within arm’s reach but out of touch; what visited the house, and where it came from? Mostly, it was moths. Between me and the moon, moth after moth. Unearthly. Of various sizes.
What I then knew about moths wouldn’t run to more than a sentence or two. Night insects, yes, the idiom “like a moth to a flame,” metamorphosis—full stop. I found a guidebook with pictures online and scrolled through it, surprised to discover that what distinguishes moths from butterflies is not, as it transpires, their circadian habit. There are many day-flying moths. Dark-loving butterfly species populate the understories of rainforests in the country’s north. Neither is the difference a question of drabness. Moths can be dotted with vivid iridescence, as if they’d dragged their wingtips through gasoline: there are green, blue, violet, pink, marigold, and piebald moths. One is the bright orange of a traffic cone; another is banana-yellow with blood-red eyeballs. In dense, low-lit woodlands, a few moth species have evolved to be almost completely transparent, a form of camouflage that means you don’t so much see the insect as notice a ripple crossing the leaf litter.
No, finer details divide moths from butterflies. Both belong to the insect order Lepidoptera, but as a general rule butterflies tend to have club-shaped antennae, where those of moths are more thready, or look like wincey bottlebrushes: an attribute that helps males pick up wafting pheromones during the breeding season. Even if they have narrow wings, moths are also more liable to have bristles on the surface between fore- and hindwing—a kind of Velcro that keeps all four wings aligned in motion—where butterflies do not. And, with exceptions, butterflies are inclined to rest holding their wings sandwiched together vertically, whereas moths idle with their wings folded over their backs, like a collapsed tent, or held out flat in the manner of a Rorschach blot.
In all its ingenuity, evolution devised a single organism capable of living two lives, at two speeds. First, the reclusive homebody, the caterpillar, a fleshy little digester in a vast empire of leaves, reliant on a plentiful if low-nutrient diet. Second, the winged moth. Extremely mobile but slight and soon to die, moths either eat nothing during their maturity or are dependent on high-energy but scarcer foodstuffs, such as sap, nectar, or juice. As a strategy, this duet of bodies has proved so successful that insects exploiting it have been around since the middle-late Jurassic. Specimens of the most primitive moths, the Micropterigidae (nine species of which live on in Australia), have been found clenched in amber from a time near to two hundred million years ago, when they might have been fodder for flying dinosaurs. With such a deep evolutionary past to pull upon, Australian moths have diversified in stable environments into a plethora of specialists, sporting an array of colors, shapes, and finishes.
Some are marbled, some are woolly. Some look like pieces of rotting wood, bird droppings, or thorns. A moth that appears to be a splotch of turquoise mold reveals startling coral-colored hindwings when it flies. Another trails streamers that baffle birds chasing it through the air. Here is a thorax as purple and shiny as plum skin; further on in the guidebook, a moth with a shaggy, bear-like countenance. Some roll their wings up to look like antlers. One seems pixelated, like a rasterized object in a video game. The smeared tones of another make it appear to have been photographed with an unfocused lens. Australia is home to between twenty and thirty thousand moth species—almost as many as there are flowering plants—but only some four hundred butterflies (a “depauperate insect fauna”).
Some moths can only be told apart by their gait when walking, having either a “waddling” gait, a “dancing gait,” or moving “quick-slow-quick” as in a foxtrot. A mothtrot. A few engage in tactical mimicry: of wasps, of repellent beetles, of less-edible moths that are their cousins. Some are furnished with hairs capable of triggering allergies and anaphylactic shock. Others would disappear if they happened to settle on an art nouveau carpet. Though the preponderance are herbivorous as larvae, there are also carnivores and frugivores. One moth tricks meat ants into carrying its caterpillars into their nests, where the larvae dine delicately on infant ants. Another has progeny that are aquatic, which subsist entirely off pondweed.
Among this spellbinding Australian bestiary are some of the world’s largest and heaviest moths. Coscinocera hercules, the Hercules moth, is found in northern Queensland and can grow to have a wingspan of thirty-six centimeters—the diameter of a car’s steering wheel. Caterpillars of the Hercules moth feed in bleeding heart trees, and then they pupate for two years. The adult moth, which moves somewhat floppily, like a dropped sunhat, lives only two days. Earlier this year construction workers sinking the foundations for a school in Mount Cotton disturbed a giant wood moth, Endoxyla cinereus, the heftiest species yet identified by science and not uncommon, though it is rarely seen. A builder balanced it on the tip of a saw for a photograph—a moth the size of a catcher’s mitt, its dusky legs dangling.
The architecture of a flower, tailored for pollination by a specific insect, can provide clues about moths unmet in the wild. Take the star-shaped orchid, from which Charles Darwin inferred the existence of a then-unknown moth with an exceptionally long tongue needed to tap the bloom’s nectary. Decades after Darwin’s death in 1882, such a moth—a subspecies of the Congo moth, X. morganii praedicta—was discovered with a ribboning proboscis almost three times the length of its body. The alliances between moths and other animals, as opposed to plants, are less well described, but as Lepidoptera elsewhere have evolved to drink the tears of river turtles, so too have unique dependencies emerged between moths and native species in Australia. There is a moth that lives in koala scat, and one that feeds only in the nests of finches. Most memorable is the moth found in holes hollowed out by golden shouldered parrots in the clay of abandoned termite mounds. There, this moth species lives off the excreta of nestlings. Though they function to keep the nest hygienic, the larvae have been known to spin cocoon masses that have blocked the entrance to the tunnel, leaving baby parrots trapped—a gothic demise for the birds.
Where once there had been hundreds of thousands of the insects—a juggernaut, a moving nimbus—now the night air stood empty.
And as for bogongs? I remembered only that they were famously innumerable and transient. Having grown up on the west coast, I had never seen one. Now I wanted to. I came to the entry for “bogong moths” in the guidebook. Agrotis infusa: the moth’s Latinate name evokes “infused fields,” a head nod to the fact that bogongs pupate mostly in croplands, in chrysalises that are sepia and translucent, like varnish on a coffin. It was the right time of year to see one. Teak brown with a fuzzy sort of cape extending over the back of its head and collar, its wings composed like a gabled rooftop when at rest. Bogongs are small, with a wingspan of about five centimeters, and they have reflective eyes—a feature that characterizes members of the Noctuidae family, those moths North Americans call “owlets” because their gleaming eyes bring to mind those of owls in the night. I read on to learn that, with the aid of a magnifying lens, it might be possible to discern that the male bogongs have antennae that resemble haircombs, though otherwise moths of this kind are unexceptional. Bogong moths are easy to miss, easy to mistake, save for this feature: on each wing are two pale dots, one slightly elongated like a comma. This is a moth adorned with semicolons.
It was the semicolons that set me off in the end: a gesture to the branching nature of sentences, and therefore of time; the possibility of subclauses running into the future, paths taken and not taken. The idea nestled into me. It was pleasant to think of something so small as a bogong moving out there, from state to state, when all else was grounded. More gratifying yet was the picture that came to me next of a stranger, their gaze alighting on a bogong moth someplace a long way away; that person becoming verily engrossed, following the moth’s mid-air helixing until it spiraled off into the dark, and then, in time, that same moth appearing to me, conveying the tiniest of contact-highs; the vision of someone somewhere else, grown watchful of the insect life nearby. Maybe there was another woman I had never met, a woman who sat by her window, even now, looking for something to wander into her reflection and bring her back to life. Perhaps she pressed her thumb against the cold surface and felt the faint vibrations of a moth squaring on the other side. If she thought of someone like me then, with the oval of her thumbprint fading, I hoped it made her feel she was not alone.
What was it that Virginia Woolf wrote, in her famous essay addressing a moth flown to the window ledge of her study? “Just as life had been strange a few minutes before, so death was now as strange.”
On the natural exodus and ingress of insects, science was, for much of history, limited to guesswork. Invertebrates have proved hard to track for all the obvious reasons—tininess, diffusion, difficulty telling kindred species apart—and, too, because many insects shapeshift across their lifecycle. Especially elusive have been “noctivagant” or night-wandering insects, those that take advantage of a drop in thermal currents after sunset to wend their way by cover of darkness. Even in very large numbers, nocturnal insects can pass by unnoticed. One morning the bugs are just here: they’re everywhere. An old magic is frisking the shrubbery. No wonder the thinkers of antiquity held that many insects were inert matter sparked to life. Aristotle and Pliny both suspected insects of being spawned by “spontaneous generation” from origins in fire, mud, dew, snow, and sand. Cicadas were fancied to issue from the spittle of cuckoos. A felicitous wind, most of all, was understood not just to transport scores of insects, but to fabricate them out of thin air.
The fact of insect migration—not to mention the basic biology of metamorphosis—has since been well substantiated, but though researchers have known for over a century that insects undertake seasonal, long-distance travel, the misguided belief that such movement is entirely passive and dictated by the winds persisted into the 1980s. Some insects clearly are at the mercy of the weather: Persectania ewingii, for example—called the “armyworm” (also, “a pest”) before it transmogrifies into a silken, buff-colored moth. Persectania ewingii is routinely tossed by spring winds from South Australia across the Bass Strait, only to be saved from perishing in the ocean by the appearance of Tasmania (or the tussock-covered shores of Macquarie Island). But as the technology capable of monitoring insects has improved, it has become clear that several winged insects—including bogongs—sense, and selectively choose, which air currents to ride, some forming massive, multispecies “bioflows” at high altitude. Radar has given entomologists a better picture of the little life gliding by far above us. What they’ve seen up there is, frankly, astounding. The spectacle of animal migration may be typified by the grandeur of herds sweeping over the Serengeti, but most terrestrial migrants are insects—by number of individuals and, perhaps more surprisingly, by mass. One study showed that each year in south-central Britain two to five trillion high-flying insects migrate over an area roughly the size of Georgia. Together, those insects have an estimated biomass greater than that of the nation’s migratory songbirds. Indeed, the volume of insects up in the air is so tremendous that researchers have suggested thinking of them as “the plankton of the sky”: a constant particulate, bobbing overhead.
At the same time our means of quantifying aerial insects has been upgraded, so too has our understanding of the impact of insect migration down on the ground. Large, traveling vertebrates, including elephants, caribou, and wildebeest, have long been known to link up ecosystems, transporting energy and nutrients (as well as pathogens) across the terrains they traverse via routine grazing, defecating, and dying. In recent years it has become clear that smaller animals, in a large enough profusion, can likewise leave a lasting impression on the landscapes they pass through. Upstream salmon rushes in British Columbia have been shown to pump nitrogen into surrounding forests, where fish carcasses fertilize fir, spruce, and cedar (so much so that tree rings record rapid growth in a banner salmon year). An animal does not need to be big to be consequential; it does not need to be warm-blooded, or a grazer, to register in the very heartwood of a forest.
The passing of mammalian herds and fish runs can score a landscape visibly by magnetizing carnivores to an area, leaving torn-up vegetation behind, or creating a “green wave” where animal activities encourage plant growth. But though the movements of insects are often more covert, over generations their transit can shape an ecosystem in equally durable ways. Very small beings have system-wide effects. Because many winged insects are pollinators, they create gene flows between plants they alight on along the route of their journey. In Spain, for instance, endangered violets surviving in geographically distant islands of habitat are genetically enchained together by the migration of hummingbird hawkmoths. Each hawkmoth threads flower to flower. Trees along a 250-kilometer stretch of the Ugab River, in Namibia, have genetic linkages that flow with the easterly movement of fig wasps. Billions of pollen grains are shifted southward each year by high-flying hoverflies in the UK, some lifted over the English Channel to the landing-pads of flowers in Europe—a targeted haulage the wind alone could never achieve. Insect migration also acts as a resource pulse in environments where endemic vertebrates feed off migrants. An influx of migrants—flies, beetles, butterflies, locusts—can intermittently decouple predator-prey dynamics, permitting prey that is low on the food chain (in many cases, local insects) to rebound, when those that eat them discover a more effortless meal of new arrivals.
Many migrating animals trail their favored weather conditions, though the bogongs must be one of only a handful of species to zero in on microclimates as pinched as those in the stony nooks they seek. To trigger aestivation, the moths need to find a protected place with a round-the-clock temperature below 16°C; a chill that must remain constant for months. In a way, the moths’ journey is not to the mountains, but to cupboards of air from a former geological age, cold air which is found during the summer now only at high elevations.
How a bogong knows where to go—how a single moth intuits the existence of a microclimate hidden in the rocks hundreds of kilometers away, far above sea level—is a perennial mystery. It cannot have been taught by its parents the way foraging bees describe routes to travel via dance: a bogong’s forebears offer no nurturance or guidance, having died before the infant moth hatches into a caterpillar. If the homing instincts of swallows and sea turtles are astonishing, consider that a bogong moth makes its journey as part of a single generation, without elders, in complete naivety. Scientists have learned that the moths can apprehend the planet’s magnetic fields and that they take their orientation from stellar cues, skills that are rare, if not unique to the moth alone, in the insect kingdom. Sometimes they fly against the wind to maintain their bearings, a determination that is especially startling when you consider how small a moth is, and how powerful the wind.
When the bogongs arrive in the Alps, mountain-living mammals rush out to gorge on moth flesh, some with pink young in the pouch. Broad-toothed rats dart about. So too Antechinus (a marsupial shrew) and the rare pygmy mountain possum. Birds binge, ravens especially. Feral pigs have developed a taste for moths. With evident cunning, the pigs remember the richest places to find them and wait there patiently for the moths to arrive, their clammy noses trembling. Nocturnal as the moths may be, the surge of bogongs up the mountainsides has been calculated to amount to an injection of energy into Australia’s alpine ecosystem that is second only to sunshine.
If the bogong moths were once called a plague, it was not because they brought sickness. Rather, their influx frightened city people with its suddenness and incessancy. A world of our design promised to restrict insect life to the margins; but having been besieged by moths, one could be forgiven for believing, however fleetingly, that our houses were less of a permanent fixture than the cascades of insects that cyclically, opportunistically, sought shelter within them. That we might have been living in the moths’ home all along proved an uncomfortable notion; one that implied we had less right to control what was excluded and what was invited in than we might have supposed. Whose world was this? Who belonged to it—and who only claimed the world belonged to them? Ideas like these were perhaps what the Belgian playwright (and armchair entomologist) Maurice Maeterlinck had in mind when, over a century ago, he wrote:
The insect brings with him something that does not seem to belong to the customs, the morale, the psychology of our globe. One would say that it comes from another planet, more monstrous, more dynamic, more insensate, more atrocious, more infernal than ours.
But then, Maeterlinck didn’t live to see that the planet “more monstrous, more dynamic . . . more atrocious, [and] more infernal” could, in fact, be our own Earth transformed. In the climate era, it is the disappearance of insects, not their efflorescence, that haunts us most of all. A global research review completed in 2019 found that 40 percent of known insect species are declining, and a third are endangered. Countless insects have suffered a reduction in range as well as numerousness—which is to say that, where they live now, many insect species also live less densely than before. In his elegiac memoir, The Moth Snowstorm (2015), British author Michael McCarthy coined the evocative phrase “the great thinning” to capture this overall loss of bug biomass. Once-common minifauna register today as exotic, hard to spot. People are more likely to assume endangered insects are interlopers, invasives, both because it is our habit to overrate our familiarity with the nature under our noses, and because those creatures that surprise us are, with increasing frequency, newcomers and novelties. Among those insects that are best able to pursue conditions beyond the boundaries of their former territory—driven along corridors of climatic change—some will be evolved by the journey so that they end up not just “out of place” but in altered bodies. (This is true of the speckled wood butterfly in Scotland, which now displays larger wing muscles along the northward edge of its habitat, where warming is drawing the butterfly into longer flights.)
Of all insect vanishings, that of the bogong moth is remarkably, terrifyingly precipitous. Insects more typically die off where habitat disappears. Those species that are most at risk are specialists with a circumscribed range and ordinarily small populations that rely on limited food plants, or are allied to threatened organisms (the rhinoceros stomach botfly can only thrive so long as there are ample rhinoceros undercarriages to parasitize). Colony insects—found in hives and anthills—are additionally vulnerable to the spread of pernicious diseases and mites because they live in such close and constant proximity to one another. As to moths particularly, traits shared by those taxa that have suffered the most rapid declines are: large wingspan, low dispersal ability, short flight seasons, and a genetic phenotype that has become “canalized” (i.e., narrowed), with fewer of the subtle perturbations seen in species that occupy multiple geographic sites.
By all these measures, the bogong moth should be secure. Insects with wings—or those that bore into, or raft upon, portable vegetable matter (picture a coconut floating in the sea)—are better able to leave a hostile environment. A few can enter into a time capsule, as those insects that burrow deep into the woody tissues of trees to pupate can sometimes wait out the barren period after a bushfire. Insects that have historically maintained very big populations have long been assumed to be comparatively insulated from extinction. Indeed, monitoring plentiful insect species not only presented technical challenges in the past but was also considered to be of low importance because models of decline held that organisms vanished at an observable rate, moving from abundant to sparse (“thinning”), and from secure to threatened, with interventions possible at several stages along the drop. What has happened to the bogong moth challenges that model of extinction and suggests that even very common insects can be at risk.
The moth’s habitat has not disappeared, taking the moth along with it. The mountains loom still; the plants the cutworms eat continue to grow in the fields (bogong larvae consume wild capeweed and crops, including cauliflower, silver beet, lucerne, flax, and other cereals). Over the decades agriculturalists have attempted to stamp out the larvae: with hellebore and pyrethrum, then with Paris green (arsenic) mixed into sweetened bran, or with a pound of DDT per acre, and by running stock and passing heavy rollers over the earth. The moths survived it all. In the last forty years, the southward expansion of rice and cotton growing in the bogongs’ natal grounds along the Murrumbidgee River, and in the Murray Valley, has no doubt curtailed their range (bogongs do not eat cotton, and the moths cannot pupate in flooded rice fields), but scientists estimate the impact of that agroeconomic shift resulted in a cull of around one-sixteenth of the population. A blow, but far from a fatal one.
What has changed, what is changing, is that the topographic features of the landscape, coded into the bogongs’ migratory instincts, are decoupling from the climatic conditions necessary for the moth to complete its life cycle. This is true at both ends of the journey. The emergence of a butterfly or moth from its pupae is called “eclosion,” and from 2017 to 2019 (and in some locations, for far longer) many of the areas where bogongs eclose were stricken by extreme drought. Temperatures climbed to highs with precedents seen only in models of a paleoclimatic hot spell two to three million years ago. The Federation Drought, the World War II Drought, even the Millennium Drought, all paled by comparison. In earth robbed of moisture, moth pupae were surely extirpated by dehydration. Larvae had less to eat when crops failed, and those robust plants that did endure were tended to more preciously by agricultural workers. The diminished number of adult moths that departed on their migration would have returned to lay eggs under those same conditions, on fields ablaze with heat.
In the mountains, too, climate change is driving an uptick in temperature. Seasonal snowmelt in the Australian Alps has inched forward almost three calendar days per decade from a baseline set in the 1950s. Mountain-living animals such as Antechinus—which breeds beneath snow cover in winter and emerges famished—now confront a metabolic rift. As the snow inches back earlier each year, surfacing females encumbered with young discover that there is little to be gleaned just when they are most in need of calories. The gap may end up being too long to bridge, when what moths there are remain on the wing kilometers away. So the changing weather is a force of disunity: the moth world and that of its predators break apart. Any life on the mountain that relies on the moths grows embrittled.
Moreover, because raised elevations are always colder than the foothills and plains, the effect of climate change on the mountains is to draw extant habitat upwards. Some plants and animals found previously in lower belts of country creep upslope towards higher altitudes, often moving into more crowded terrain (as the surface area of a peak narrows into a summit). Organisms found at the tops of mountains face the evaporation of their domain into midair when apex conditions exceed their tolerance. For the bogong moths this means that the coldest crevices are today found closer to the summit, while their ancestral bolt-holes below grow increasingly unsuited to aestivation. The day may yet come when those moths that are left fail to find the microclimate they need to rest. So the survival of the moths rests not just on the mountains, but on the atmosphere in which the mountains stand.
The vanishing of the bogong moth betrays an invisible and global threat, yet it is because the moths were once so abundant that their loss is palpable to us. That multiple populations of moths coalesced in the mountains, rather than remaining dispersed over a vast landmass, served to dramatize their absence. Empty caves are an image the mind can readily latch onto. The fact that the bogongs once proliferated in places of work and residence, too close for comfort, also made them memorable. Would their disappearance have been overlooked were they a less numerous, more cryptic species?
What do we stand to lose when we lose the moth? The answer is many-layered. If their worth is as a traction, joining worlds, then migratory bogongs need to be preserved in abundance. From an evolutionary standpoint, moving as a multitude permits an insect species to glut those predators it cannot evade and to absorb losses owing to adversities like bad weather. With no intrinsic defense (beyond camouflage), each bogong moth’s safekeeping owes to the company of many more moths than appetite or attrition can account for. Beyond a minimum threshold, the moth gains new vulnerabilities. Some kindred insect species appear to require a set population size and density to be reached—a quorum—before they are pushed off whatever inner ledge holds them in place and start their migration. What liberates these insects from stasis is not intention, or a move to action, but surrender to the vigor of amassing together. Dwindling can therefore be a force like inertia. Is it absurd to imagine that, for insects and other animals, being deprived of migration has an emotional dimension? Between captivity and freedom is there an apprehension of restraint, of capture in a sphere smaller yet than desire fills? What is the name for this grief? Could it inhabit something so small as a moth? A few secluded populations of bogongs don’t migrate. The non-migratory morph has paler hindwings and completes its lifecycle in a different part of the calendar than its much more prolific, wayfaring counterpart: a wan, agoraphobic sibling.
What is the name for this grief? Could it inhabit something so small as a moth?
Abundance matters, too, because rarity renders the migration’s knock-on effects negligible or defunct: the migration’s ecological consequences crumble where few moths undertake it. Though we customarily speak of needing to save a habitat to save a species, per those animals that move in vast numbers the opposite is also true. The preservation of habitat, its energetic balance, pivots on the transient animals that pass through it. A depletion of migratory animals can be a force as atrophying as plunging the land into darkness.
The bogong moth itself may not be beautiful, but it is a cornerstone of the beauty found in the mountains where the moths pollinate flowers and nourish alpine biomes. And yet, there is more at stake here than beauty. Upland ecosystems have downstream effects. The Alps are sometimes referred to as Australia’s “liquid lungs” for their watershed function and because they filter rain running into aquifers that supply the cities. If the bogong moth, a keystone species, does not recover, the tattering of mountain nutrient cycles may reduce water quality elsewhere.
To argue for shielding animal migration is to adopt an expansive definition of vulnerability, for it means not just protecting animals—safeguarding their mere existence—but maintaining their ways of being in the world, and in relationship to one another. It can also mean attending to the commonplace over the seldom-seen; to the maintenance of the unremarkable. Yet only the most cold-eyed ecologist would view an animal’s worth as exclusively a matter of tabulating benefit to its surroundings, dependents, and consumers. The moths don’t just connect ecosystem to ecosystem, they connect people to people, and people to the past. Indigenous owners of the moth aestivation grounds are cognizant of the moth as a talisman for the Country it enlivens, and of its pathway through the world as a more durable kind of knowledge than anything illuminated by data. One thing we all stand to lose if the bogong moth disappears for good is a feeling for our own smallness and impermanence—for even as each moth is turned about in less than a year, the flow it belongs to is ancient.
On a morning not long ago, as the city began to stir out of its own long inertia, I drove to Melbourne Zoo to visit the Butterfly House. In the queue, corralled along the zoo’s perimeter, people fumbled to pull up vaccine passports on the government app, as zoo workers in khakis implored ticket holders to observe social distancing by keeping “the length of a kangaroo” between family groups: an interval soon collapsed by impatience. Children whooped up and down the line while mothers traded glances of bone-weary camaraderie. As the clouds flew off overhead and the temperature began to climb, I wondered if the animals penned inside heard the hum of the crowd about to descend and whether that noise aroused anxiety or anticipation in them. Had any of the zoo’s creatures worried over where their spectators had gone and why they had disappeared for so long?
The Butterfly House is steamy, with rings of rock melon on wire suspended from the trees and hexagonal feeding tables set with plastic florets, where sugar water is set out. Around 450 Lepidoptera quiver on vegetation, or on the feeding tables, and thresh in the air. Almost all are butterflies, though there is one day-flying moth species among them—the Hercules moth, a few saucer-sized individuals very far from the tropics where they usually make their home. On the concertina of a palm leaf, two orchard swallowtails look to be mating, the fainter female like a sandy shadow beneath the male.
There are no bogongs. To date, I have not seen one. So far this year, the mountains have not either. Yet it is hard to put into words the upswing of emotion I experience in the Butterfly House nonetheless. I can’t help it. To be surrounded by so much life this fragile and ornate is overwhelming. An older man is frozen mid-step by a butterfly alighting on his forehead, on the spot notionally known as the third eye. A bygone name for Lepidoptera is psykhe (psychê), a term that later denoted the soul. For a moment, I also stop to take stock of a feeling inside me. Something moth-sized has taken flight within my body, a ricocheting brightness with an autonomy all its own, a lightness that had eluded me all through the long second half of the year. As though these vivid insects, their presence, have broken something heavy within me into parts and made it available to catch on a breeze.
Bogong moths are not social insects. Social insects, such as honeybees and termites, have a hierarchy and distribute roles to different individuals. What the moths are is massively gregarious—and what they are losing now is their coming together. Denied to the moths is the momentum to densify, to persevere high in the overworld, and endure ever more infernal summers by resting, wingtip to wingtip. And yet, some of the planet’s most isolated insects have been restored by human effort. The Butterfly House ordinarily displays one non-Lepidoptera insect at intervals, in a terrarium brought out from enclosures not open to the public: the Lord Howe Island stick insect, a bug also called “the tree lobster” after its size. Considered extinct since 1920, a breeding colony of just twenty-four Lord Howe Island stick insects was discovered in 2001 clinging to the ground beneath a single shrub on a mid-ocean crag of rock, a sea mountain called Ball’s Pyramid, in the Pacific Ocean, over six hundred kilometers northeast of Sydney. The two stick insects (the zoo named them Adam and Eve), brought to Melbourne by their discoverers, have since sired over fourteen thousand offspring, though I’m told they are not on display today because people too easily forget the social distancing protocols when they gather around to view them.
Are we in time to double back, to save the bogongs? The revival of the moth won’t be so easy to secure. On Lord Howe Island, the stick insects were killed off by invasive black rats introduced by shipping vessels: a localized catastrophe. The plight of the bogongs, on the other hand, is entangled with the global problem of climate change. All migratory species depend on a series of habitats, sequenced conditions in those habitats, and the transitional spaces that provide passage between them. Such species are contingent on a far greater domain, replete with many more resources, than is needed to supply the individual. A single insect may live off a tablespoon of nutriment. A dozen may occupy a shoebox without obvious antipathy. And though a bogong could survive in the Butterfly House, the fate of its species hinges on weather patterns that encompass the globe. And yet, hope resides in the insect. Having evolved to produce hundreds of offspring (over a strategy of cosseting a few), each generation has the potential for mass depletion, but also replenishment. One adult can lay two thousand eggs. In the wake of the IUCN listing, conservation advocates are now pushing for improved moth habitat within agricultural lands, offset by subsidies similar to those offered to some EU farmers for engaging in bee-friendly growing methods. Some reprieve also looks set to arrive with the shift into a La Niña season, historically wetter and cooler on the eastern side of Australia.
My gaze returns to the butterflies when one lands on the back of my hand and pauses there, closing and unclosing like a pamphlet of inscrutable information. A voice piped over the speakers warns visitors not to try to stroke the insects or grab for them. Even with the greatest of care, it says, our touch can hurt them.